Laméris is a specialist supplier for the ophthalmic practice. From advice to service! They saw that there was a void in orthopedic equipment. Orthotics is concerned with diagnosing the musculoskeletal system of the eye, with the best example being strabismus. After a study carried out at the VU medical center in Amsterdam, the idea for the NINE was born.
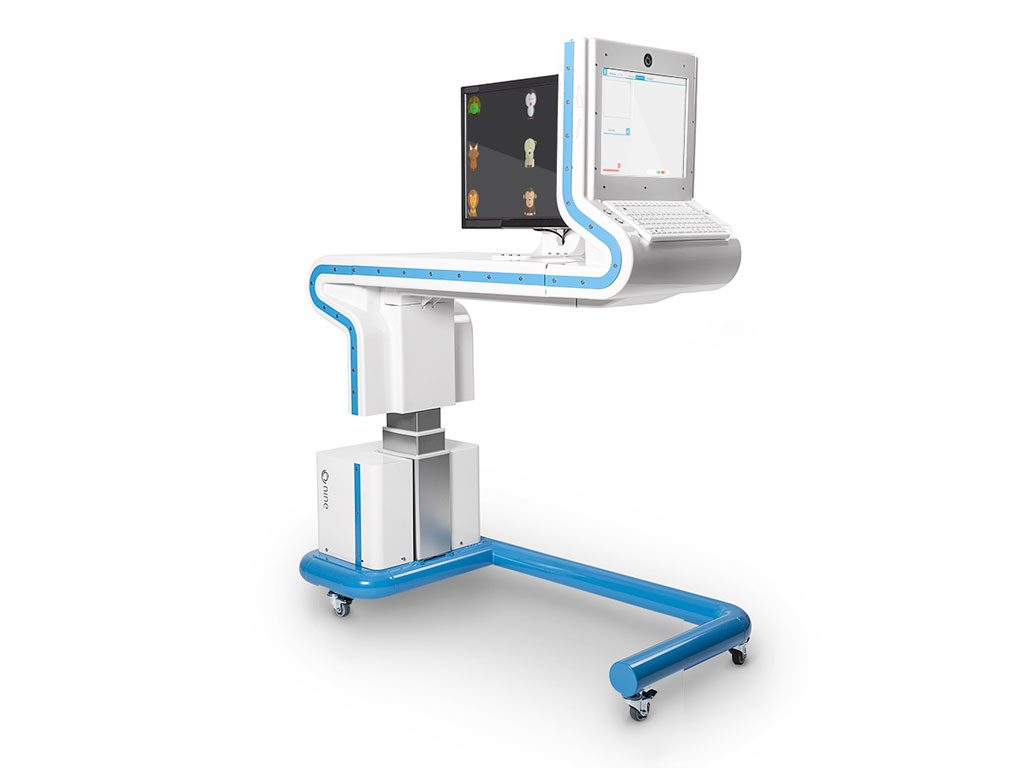
NINE stands for 9 directions to look for for the particular eye exam. The device stimulates the patient to look at the different directions. This is done by offering a game on a screen. While running this game, the eye movements are tracked and analysis is performed.
R2Pro has been asked to translate the LAB model of the VU Medical Center into a salable end product. We did this in full collaboration with the Orthoptists of the VU medical center, so that they also had the feeling that the device was developed for them. Laméris Ootech subsequently opted for the revolutionary concept in which the device and table are integrated and optimally suited to patients and orthoptists.
- Extensive user research in hospitals
- Elaboration of multiple concepts, mechanical, electronic, software
- Concepts were assessed by users and presented to Laméris Ootech with these results.
- Searching and collaborating with various suppliers.
- Maintain necessary documentation for medical regulations.
- Development of the product into a prototype that was shown at the International Orthoptic Congress